
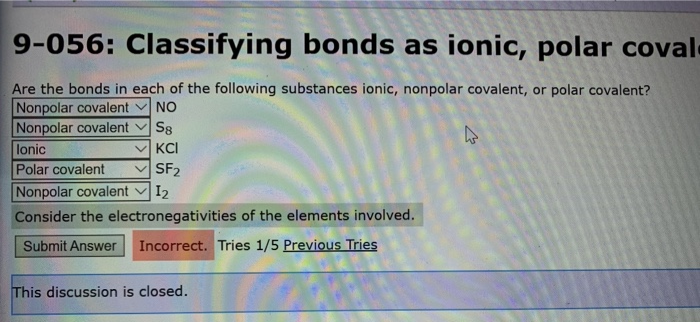
In this type of interaction, a non-polar molecule is polarized by an ion placed near it. The molecular mass of the SF2 molecule is 70.062 g/mol. Answer (1 of 6): To figure out whether a molecule is polar or not, you have to consider the dipole moments of each contributing bond in the molecule, and then decide whether these dipole moments are exactly cancelled out, or if there is a resultant dipole from adding together all of the individ. Size of the polar molecule The size and charge of an ion 3. Three monosulfur fluorides are observed: SF2, SF4, and SF6.

When the physique is below assault from organisms (e.g. It is a polar molecule and a three death each of the saved and the DJ polar molecule E five. A molecule may be nonpolar either when there is an equal sharing of electrons between the two atoms of a diatomic molecule or because of the symmetrical arrangement of polar bonds in a more complex molecule. Therefore, the dipoles of the S-F bond do not cancel out each other and molecules turn out to be polar and contribute some dipole moment. Draw Lewis structures, name shapes and indicate polar or non-polar for the following molecules: a.
